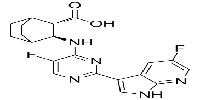
-
匹莫迪韋
- names:
Pimodivir
- CAS號:
1629869-44-8
MDL Number: MFCD31382121 - MF(分子式): C20H19F2N5O2 MW(分子量): 399.39
- EINECS: Reaxys Number:
- Pubchem ID:67286591 Brand:BIOFOUNT
| 貨品編碼 | 規格 | 純度 | 價格 (¥) | 現價(¥) | 特價(¥) | 庫存描述 | 數量 | 總計 (¥) |
|---|---|---|---|---|---|---|---|---|
| YZM000729-5mg | 5mg | 99.04% | ¥ 1569.00 | ¥ 1569.00 | 2-3天 | ¥ 0.00 | ||
| YZM000729-2mg | 2mg | 99.04% | ¥ 965.25 | ¥ 965.25 | 2-3天 | ¥ 0.00 |
| 中文別名 | 匹莫迪韋(1629869-44-8);吡莫地韋;VX-787;VX 787;VX787; JNJ-872;JNJ 872;JNJ872;VRT-0928787; VRT 0928787;VRT0928787;吡莫地爾; (2S,3S)-3-((5-氟-2-(5-氟-1H-吡咯并[2,3-b]吡啶-3-基)嘧啶-4-基)氨基)雙環[2.2.2 ]辛烷-2-羧酸; |
| 英文別名 | Pimodivir(1629869-44-8);VX-787;VX-787; VX 787; VX787; JNJ-872; JNJ 872; JNJ872; VRT-0928787; VRT 0928787; VRT0928787;pimodivir; (2S,3S)-3-((5-Fluoro-2-(5-fluoro-1H-pyrrolo[2,3-b]pyridin-3-yl)pyrimidin-4-yl)amino)bicyclo[2.2.2]octane-2-carboxylic Acid; |
| CAS號 | 1629869-44-8 |
| Inchi | InChI=1S/C20H19F2N5O2/c21-11-5-12-13(7-24-17(12)23-6-11)18-25-8-14(22)19(27-18)26-16-10-3-1-9(2-4-10)15(16)20(28)29/h5-10,15-16H,1-4H2,(H,23,24)(H,28,29)(H,25,26,27)/t9?,10?,15-,16-/m0/s1 |
| InchiKey | JGPXDNKSIXAZEQ-SBBZOCNPSA-N |
| 分子式 Formula | C20H19F2N5O2 |
| 分子量 Molecular Weight | 399.39 |
| 溶解度Solubility | 生物體外In Vitro:DMSO溶解度100 mg/mL(250.38 mM;Need ultrasonic) |
| 性狀 | 固體粉末,Power |
| 儲藏條件 Storage conditions | 存放在陰涼干燥處,短期(數天至數周)在0-4°C下,長期(數月至數年)在-20°C下保存。 |
匹莫迪韋(1629869-44-8,Pimodivir,VX-787)實驗注意事項:
1.實驗前需戴好防護眼鏡,穿戴防護服和口罩,佩戴手套,避免與皮膚接觸。
2.實驗過程中如遇到有毒或者刺激性物質及有害物質產生,必要時實驗操作需要手套箱內完成以免對實驗人員造成傷害
3.實驗后產生的廢棄物需分類存儲,并交于專業生物廢氣物處理公司處理,以免造成環境污染Experimental considerations:
1. Wear protective glasses, protective clothing and masks, gloves, and avoid contact with the skin during the experiment.
2. The waste generated after the experiment needs to be stored separately, and handed over to a professional biological waste gas treatment company to avoid environmental pollution.
Tags:匹莫迪韋試劑,匹莫迪韋雜質,匹莫迪韋中間體,匹莫迪韋密度,匹莫迪韋純度,匹莫迪韋閃點,匹莫迪韋熔點,匹莫迪韋合成,匹莫迪韋溶解度,匹莫迪韋購買,
| 產品說明 | 匹莫迪韋(1629869-44-8,Pimodivir,VX-787)是一種有效的可口服的甲型流感病毒聚合酶的抑制劑,通過抑制PB2亞基起作用 |
| Introduction | 匹莫迪韋(1629869-44-8,Pimodivir,VX-787)is an orally bioavailable inhibitor of influenza A virus polymerasesthrough interaction with the viralPB2subunit. |
| Application1 | Pimodivir(VX-787)是一種可口服的甲型流感病毒聚合酶抑制劑,通過抑制PB2亞基作用。 |
| Application2 | 已發現吡咯并吡啶衍生物VX-787是一種抗病毒藥,目前仍處于針對甲型流感病毒感染的II期試驗中。 |
| Application3 |
Pimodivir是A型流感病毒聚合酶復合物的聚合酶基礎蛋白2(PB2)亞基的口服生物可利用的非核苷抑制劑,具有潛在的抗病毒活性。給藥后,匹莫地韋占據PB2 的7-甲基GTP(m7GTP)結合位點,從而阻斷流感聚合酶復合物的奪帽活性并抑制病毒mRNA的合成。PB2是組成甲型流感病毒聚合酶復合物的三個亞基之一,它負責感染細胞核中病毒RNA(vRNA)基因組的復制和轉錄。
| 警示圖 | |
| 危險性 | warning |
| 危險性警示 | Not available |
| 安全聲明 | H303吞入可能有害+H313皮膚接觸可能有害+H2413吸入可能對身體有害 |
| 安全防護 | P264處理后徹底清洗+P280戴防護手套/穿防護服/戴防護眼罩/戴防護面具+P305如果進入眼睛+P351用水小心沖洗幾分鐘+P338取出隱形眼鏡(如果有)并且易于操作,繼續沖洗+P337如果眼睛刺激持續+P2393獲得醫療建議/護理 |
| 備注 | 實驗過程中防止吸入、食入,做好安全防護 |
| Smee DF, et al. Activities of JNJ63623872 and GS 4071 against influenza A H1N1pdm and H3N2 virus infections in mice. Antiviral Res. 2016 Dec;136:45-50. |
| Fu Y, et al. JNJ872 inhibits influenza A virus replication without altering cellular antiviral responses. Antiviral Res. 2016 Sep;133:23-31. |
| Boyd MJ, et al. Isosteric replacements of the carboxylic acid of drug candidate VX-787: Effect of charge on antiviral potency and kinase activity of azaindole-based influenza PB2 inhibitors. Bioorg Me |
| Byrn RA, et al. Preclinical activity of VX-787, a first-in-class, orally bioavailable inhibitor of the influenza virus polymerase PB2 subunit. Antimicrob Agents Chemother. 2015 Mar;59(3):1569-82. |
| A Phase 2 Study of Pimodivir (JNJ-63623872) in Combination with Oseltamivir in Elderly and NonElderly Adults Hospitalized with Influenza A Infection: OPAL study PMID 32604406; The Journal of infectiou |
匹莫迪韋(1629869-44-8,Pimodivir,VX-787)參考文獻:
1.Preclinical activity of VX-787, a first-in-class, orally bioavailable inhibitor of the influenza virus polymerase PB2 subunit.
Byrn RA;Jones SM;Bennett HB;Bral C;Clark MP;Jacobs MD;Kwong AD;Ledeboer MW;Leeman JR;McNeil CF;Murcko MA;Nezami A;Perola E;Rijnbrand R;Saxena K;Tsai AW;Zhou Y;Charifson PS Antimicrob Agents Chemother. 2015 Mar;59(3):1569-82. doi: 10.1128/AAC.04623-14. Epub 2014 Dec 29.
VX-787 is a novel inhibitor of influenza virus replication that blocks the PB2 cap-snatching activity of the influenza viral polymerase complex. Viral genetics and X-ray crystallography studies provide support for the idea that VX-787 occupies the 7-methyl GTP (m(7)GTP) cap-binding site of PB2. VX-787 binds the cap-binding domain of the PB2 subunit with a KD (dissociation constant) of 24 nM as determined by isothermal titration calorimetry (ITC). The cell-based EC50 (the concentration of compound that ensures 50% cell viability of an uninfected control) for VX-787 is 1.6 nM in a cytopathic effect (CPE) assay, with a similar EC50 in a viral RNA replication assay. VX-787 is active against a diverse panel of influenza A virus strains, including H1N1pdm09 and H5N1 strains, as well as strains with reduced susceptibility to neuraminidase inhibitors (NAIs). VX-787 was highly efficacious in both prophylaxis and treatment models of mouse influenza and was superior to the neuraminidase inhibitor, oseltamivir, including in delayed-start-to-treat experiments, with 100% survival at up to 96 h postinfection and partial survival in groups where the initiation of therapy was delayed up to 120 h postinfection.
2.Pimodivir treatment in adult volunteers experimentally inoculated with live influenza virus: a Phase IIa, randomized, double-blind, placebo-controlled study.
Trevejo JM;Asmal M;Vingerhoets J;Polo R;Robertson S;Jiang Y;Kieffer TL;Leopold L Antivir Ther. 2017 Dec 15. doi: 10.3851/IMP3212. [Epub ahead of print]
BACKGROUND: ;Pimodivir (formerly JNJ-63623872) is a novel, non-nucleoside polymerase complex inhibitor with in vitro activity against influenza A virus, including pandemic 2009 H1N1, H7N9, H5N1 strains as well as neuraminidase- and amantadine-resistant strains.;METHODS: ;Randomized, double-blind, placebo-controlled, Phase IIa study. Healthy volunteers (n=104) were inoculated with an influenza A/Wisconsin/67/2005 (H3N2) challenge virus. 72 received pimodivir and 32 placebo. Pimodivir was dosed for 5 days once daily from 24 h after viral inoculation at four dose levels: 100 mg, 400 mg, loading dose 900/600 mg and loading dose 1,200/600 mg.;RESULTS: ;Pimodivir significantly reduced viral shedding (area under the concentration versus time curve [AUC] measured by 50% tissue culture infective dose [TCID;50;] or qRT-PCR) versus placebo as measured by cell culture assay in the pooled analysis (Jonckheere-Terpstra dose-response trend test [P=0.036]). Reductions were observed in viral shedding (AUC, duration and peak measured by grade), influenza-like symptoms (AUC, duration and peak measured by grade) and clinical symptoms (duration and peak measured by grade) for all pimodivir groups versus placebo, significantly so for the 1,200/600 mg group.
3.JNJ872 inhibits influenza A virus replication without altering cellular antiviral responses.
Fu Y;Gaelings L;Söderholm S;Belanov S;Nandania J;Nyman TA;Matikainen S;Anders S;Velagapudi V;Kainov DE Antiviral Res. 2016 Sep;133:23-31. doi: 10.1016/j.antiviral.2016.07.008. Epub 2016 Jul 20.
JNJ-63623872 (formally known as VX-787; referred to here as JNJ872) is an orally bioavailable compound, which is in phase II clinical trials for the treatment of influenza A virus (IAV) infections. Here we show that JNJ872 inhibits at nanomolar concentrations the transcription of viral RNA in IAV-infected human macrophages by targeting a highly conserved site on the cap-snatching domain of influenza polymerase basic 2 protein (PB2). Furthermore, even lower concentrations of JNJ872 protected macrophages from IAV-mediated death when given in combination with 100 nM gemcitabine, which also attenuated transcription and replication of viral RNA. Importantly, treating human macrophages with JNJ872 allowed expression of many immune-related and other genes, involved in antiviral responses, such as indoleamine 2,3-dioxygenase 1 (IDO), and cytosolic 5'-nucleotidase 3A (NT5C3A). Moreover, our targeted metabolomics analysis indicate that treatment with JNJ782 did not interfere with metabolic responses to infection, which further supported our transcriptomics results. Thus, VX-737 alone or in combination with other drugs could be beneficial for treating IAV infected patients, because it would allow the development of antiviral responses and, thereby, protect individuals from current and future infections with closely related IAV strains.
- 相關產品
-
< >
- 推薦產品
-
< >
- 最新產品
-
< >
新聞

怎么做細胞爬片免疫組化染色實驗
細胞爬片免疫組化染色,是通過細胞爬片是讓玻片浸在細胞培養基內,細胞在玻片上生長,主要用于組織學,免疫組織化學...
2020/7/20 22:04:33
提取病毒RNA的實驗方法
提取病毒RNA方法分別有:異硫氰酸胍的提取病毒RNA方法、TRIzol LS提取法、Trizol法提取法等等...
2020/7/22 20:29:26

chelex 100樹脂國產替代之路-BIOFOUNT范德生物
Chelex 100螯合離子交換樹脂對銅、鐵和其他重金屬?的偏好顯著高于對鈉、鉀等一價陽離子的偏好。它對二價...
2025/11/4 14:22:46

9月開學季——助研新學期 范德送好禮
2025/8/28 15:30:55

Waxfilm 實驗室封口膜:技術與國際市場的雙重突破
在實驗室耗材領域,封口膜是保障實驗準確性與穩定性的關鍵產品之一。近年來,Waxfilm?實驗室封口膜憑借其卓...
2025/5/13 13:03:40

Waxfilm實驗室封口膜的5大突破
Waxfilm實驗室封口膜作為生物功能膜領域的國產技術突破和品牌突破,是生物領域中國技術發展的縮影。
2025/5/6 17:02:07

各種微流控芯片鍵合方法的優缺點
微流控芯片鍵合:目前主要有激光焊接、熱壓鍵合、膠鍵合、超音波焊接,每種方法都有各自的優缺點。本文主要介紹聚酯...
2023/7/28 10:43:09

新一代微流控鍵合解決方案
微流控鍵合解決方案:微流控芯片制造的一個重要環節,也是最容易被忽視的--芯片鍵合。其中一個重要因素是:微流控...
2023/7/27 12:44:28

熒光素鉀鹽使用說明
D-熒光素鉀鹽(K+)設計用于體外和體內生物發光測定。D-熒光素的質量和純度對于獲得良好和可重復的結果至關重...
2023/7/20 11:05:11
如何選BSA(牛血清白蛋白)
如何選BSA(牛血清白蛋白):牛血清白蛋白(BSA)有多種形式,如何選擇適合自己的牛血清白蛋白(BSA)是一...
2023/2/14 13:09:18




 購物車
購物車 



